What alkenes would give only an aldehyde with four carbons as a product of oxidative cleavage?
1 Answer
Jan 13, 2015
The six possible alkenes are the cis and trans isomers of oct-4-ene, 2,5-dimethylhex-3-ene, and 1,2-di(cyclopropyl)ethene.
Oxidative cleavage of an alkene is the conversion of the alkene carbons into separate carbonyl groups.

We can work backwards from the products and figure out what the alkene must have been.
If the product was RCHO, the alkene must have been RCH=CHR.
There are three 4-carbon aldehydes:
Butanal

2-Methylpropanal

Cyclopropanecarboxaldehyde

From these, we can work backwards and say that the starting material must have been the E or Z isomer of
Oct-4-ene

2,5-Dimethylhex-3-ene
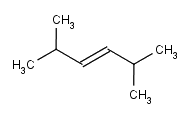
or
1,2-Di(cyclopropyl)ethane

So there are six possible alkenes that give only a four-carbon aldehyde by alkene cleavage.

