What is oxidative cleavage?
1 Answer
Oxidative cleavage is the cleavage of carbon-carbon bonds to generate carbon-oxygen bonds. Sometimes
OXIDATIVE CLEAVAGE: PERIODIC ACID
In general, an oxidative cleavage of a vicinal diol (commonly with periodic acid---pronounced "per-iodic") looks like this:
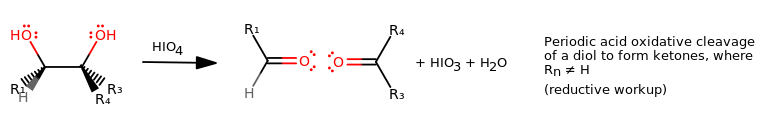
Notice how the
PERIODIC ACID OXIDATIVE CLEAVAGE MECHANISM
The mechanism goes as follows (ignore the red iodine; the valency ought to be fine):
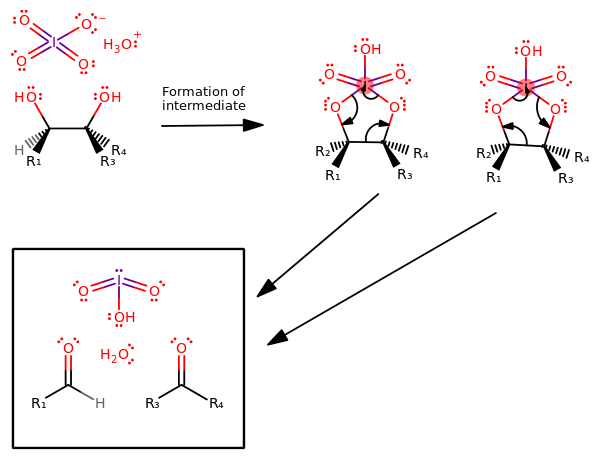
You don't have to know the mechanism, but I think it helps to have some idea on what happens "behind the scenes".
OXIDATIVE CLEAVAGE: OZONE (ALKENE)
On the other hand, an oxidative cleavage of an alkene using ozone looks like this:
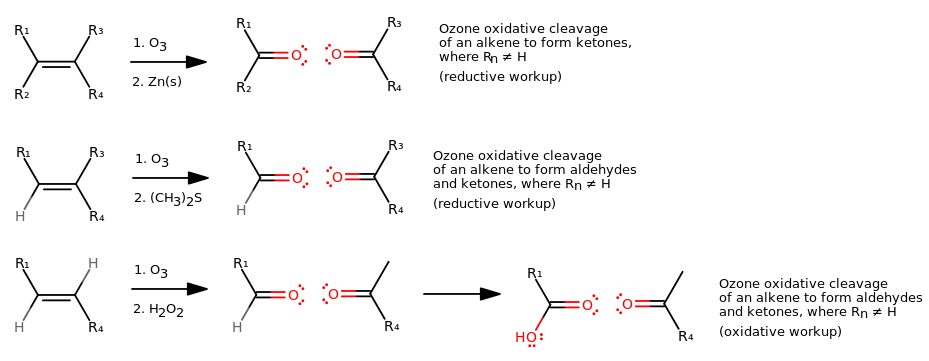
For the first step, notice how in essence, the
REDUCTIVE WORKUP
For the second step, adding
OXIDATIVE WORKUP
For the second step, adding
Instead of stopping at the aldehyde or ketone, you go all the way to a carboxylic acid or ketone.
OZONE OXIDATIVE CLEAVAGE MECHANISM (ALKENE)
The general mechanism for oxidative cleavage of an alkene using ozone is:
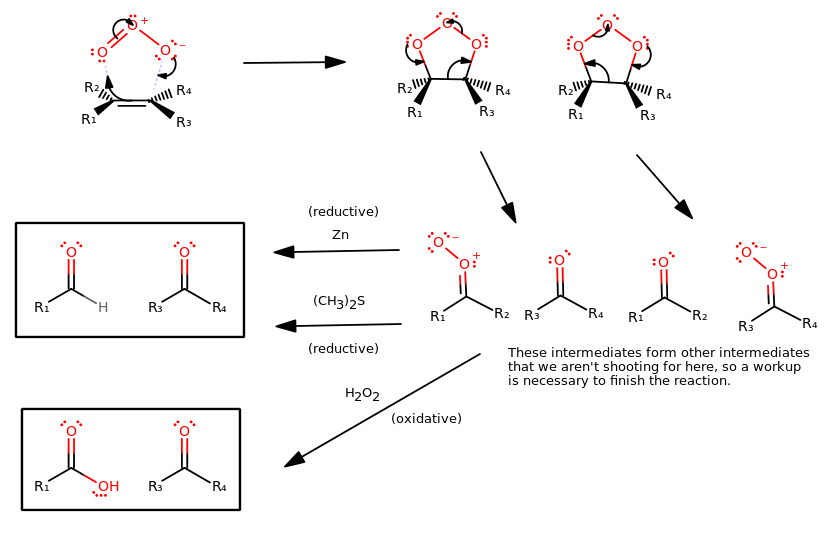
OXIDATIVE CLEAVAGE: OZONE (ALKYNE)
For an alkyne:
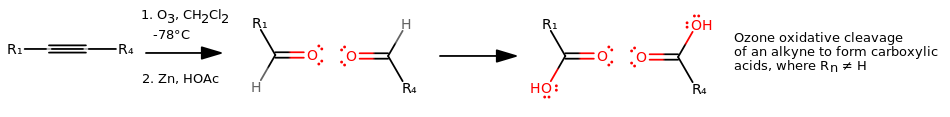
OZONE OXIDATIVE CLEAVAGE MECHANISM (ALKYNE)
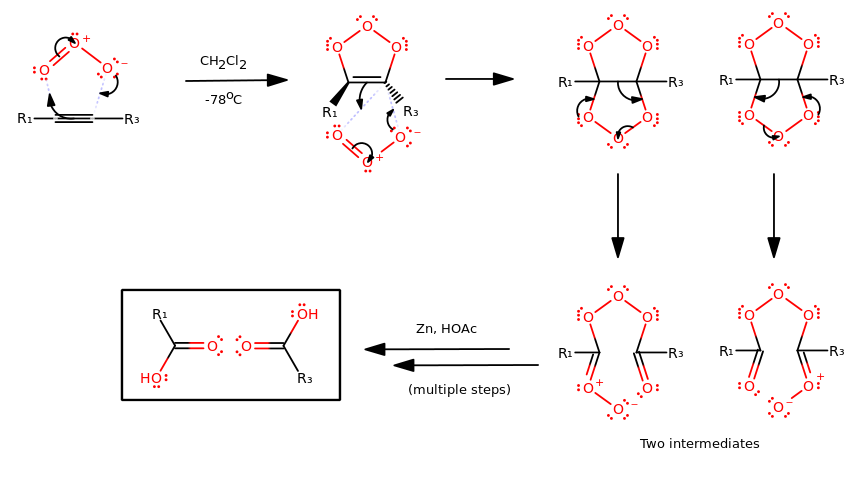
You don't have to know either mechanism, but I think it helps to have some idea on what happens "behind the scenes".

