What are some examples of bonding orbitals?
1 Answer
May 8, 2015
The
Another example is a
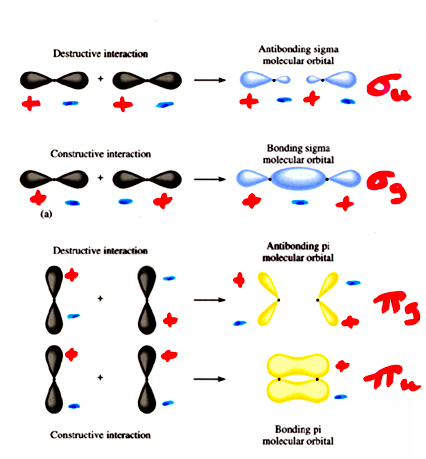
Look at the "Antibonding sigma molecular orbital" for the
The
Another example is a

Look at the "Antibonding sigma molecular orbital" for the