What are the optical isomers of amino acids?
1 Answer
Dec 20, 2016
They are the two non-superimposable mirror image isomers that all alpha-amino acids except glycine have.
Explanation:
Alpha- amino acids have a central carbon atom with a tetrahedral arrangements of bonds around it, to which
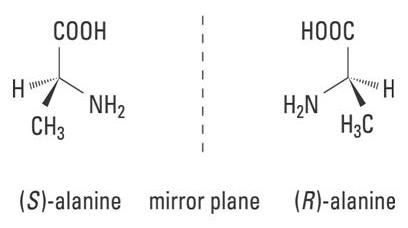
Diagram source: http://www.rowland.harvard.edu/rjf/fischer/background.php
A carbon such as this central one is referred to as chiral. A more complex molecule may have more than one chiral centre, and therefore more than two optical isomers.

