What force holds two atoms together?
1 Answer
Jan 5, 2017
The force of electrostatic attraction...........
Explanation:
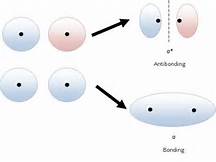
The modern chemical bond is conceived to be a region of high electron density between 2 positively charged atomic nuclei such that nucleus-nucleus repulsion is negated, and a net attractive force results.
A bonding orbital, which can accommodate 2 electrons, is conceived to be that region of high electron density, and our mathematical representations of how electron density is distributed shows the concentration of charge, i.e. electrons, in the region between the atoms.
See here: