What is the conjugate acid of ammonia that forms when it is added to water?
1 Answer
Ammonium,
Explanation:
Conjugate acid-base pairs consist of two compounds, differing by a charge of +/-1 due to the movement of a single proton
You use the bronstead description of acids and bases when calculating which is the conjugate acid and which is the conjugate base in a reaction. This theory explains that a bronstead acid is any molecule, ion or atom that can donate a proton and a bronstead base is any molecule, ion or atom that can accept a proton.
In this reaction, ammonia is the conjugate base and accepts a proton from water, where it becomes the conjugate acid ammonium. You can show that the lone pair of electrons on the nitrogen atom reacts with the hydrogen on water molecules, giving an ammonium ion and a hydroxide ion.
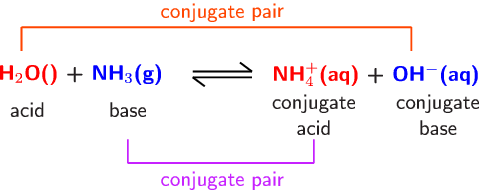
An extra resource that might help:
https://www.khanacademy.org/science/chemistry/acids-and-bases-topic/copy-of-acid-base-equilibria/v/conjugate-acid-base-pairs-acids-and-bases-chemistry-khan-academy
I hope this helps
Slothdemon

