What is the correct electron configuration for silicon?
2 Answers
Explanation:
When adding electrons, the lowest energy levels are always filled first. This is shown by the Aufbau princible shown here:
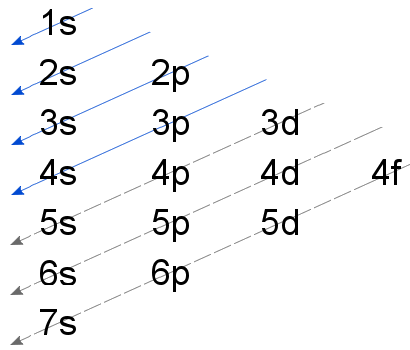
The lowest energy level is the 1s. All orbitals hold two electrons, and there is one possible orbital for s electrons to have, so we add two electrons to 1s. The same is true for the next highest level, 2s. 2p is different because there are three different orbitals which can hold 6 electrons in total. The same filling continues, and you add two electrons to the 3s. At this point, we have used twelve electrons and two electrons remain since silicon has 14 electrons. We add the final two electrons to the 3p. So the final configurations is:
Explanation:
Although the alternate explanation is correct by @tcw1 if looking at a simpler level...
An electronic configuration describes how many electrons are in an elements shells. Depending on how many electrons an element has the number of shells and electronic configuration can differ.
For lower level, the electronic configuration goes to a maximum of
We can work out the electronic configuration from the periodic table.
Silicon is the
Since elements can only have up to
Since the
The third shell can hold up to


