What is the electron configuration for a phosphide ion?
1 Answer
Explanation:
The electron configuration of phosphorus is
When the phosphide ion forms, the phosphorus atom gains three electrons. Thus, the electron configuration becomes
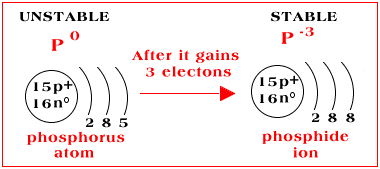
The electron configuration of phosphorus is
When the phosphide ion forms, the phosphorus atom gains three electrons. Thus, the electron configuration becomes
