What is the electron configuration for As?
1 Answer
The complete electron for a neutral arsenic atom is:
Its shorthand electron configuration is:
Explanation:
As is the chemical symbol for the element arsenic. Its atomic number is 33, which is the number of protons in the nuclei of its atoms. In a neutral atom, the number of electrons equals the number of protons, which means that neutral arsenic atoms have 33 electrons.
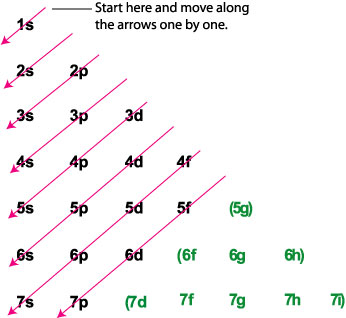
By using the Aufbau diagram (shown below) we can determine the full electron configuration for a neutral arsenic atom.
The shorthand electron configuration, which uses the symbol for the noble gas in the previous period, in this case argon.
Aufbau Diagram