What is the electron configuration of Li+?
1 Answer
Explanation:
Your starting point here will be the electron configuration of a neutral lithium atom,
A quick look in the periodic table will reveal that lithium is located in period 2, group 1, and that it has an atomic number equal to
This means that a neutral lithium atom will have a total of
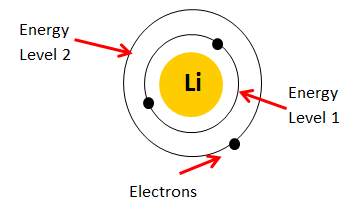
Its electron configuration will be
Li: 1s22s1
Now, the lithium cation,
This means that the electron configuration of the
Li+:1s2
To write this using noble gas shorthand notation, use the electron configuration of the noble gas that comes before lithium in the periodic table.
Helium,
He: 1s2
This means that you have
Li+:[He]
Here the notation

