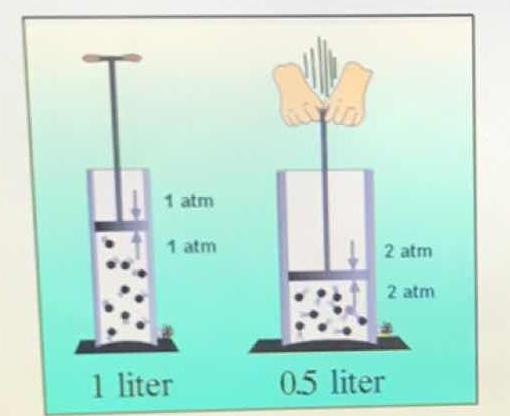
What gas law is illustrated by this picture?
1 Answer
Jun 7, 2017
Explanation:
Boyle's Law states that pressure and volume of a gas are inversely proportional (
As the piston is lowered to a new volume


