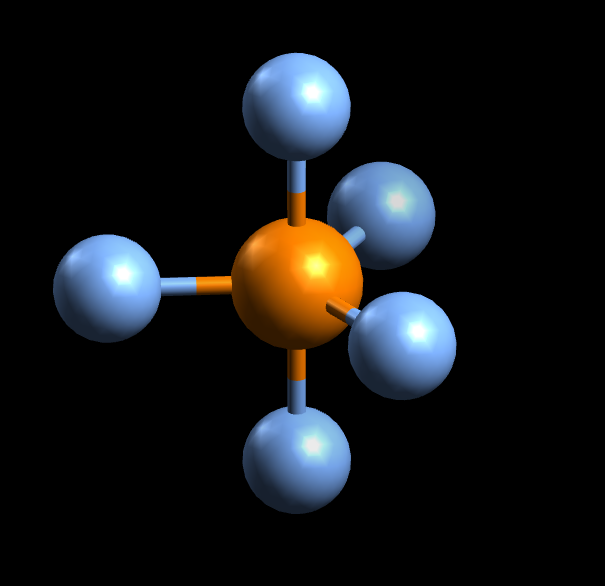
#PBr_5#'s molecular geometry is trigonal bipyramidal.
Let's start with its Lewis structure. #P# has 5 valence electrons and #Br# has 7 valence electrons, which means that a total of #5 + 5*7 = 40# electrons must be accounted for.
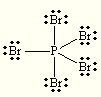
#P#'s bonds with the five #Br# atoms account for #5 * 2 = 10# electrons, while the three lone pairs on each #Br# atom complete the rest of the electrons - #5 * 6 =30# electrons.
SInce #P# forms bonds with 5 other atoms and has no lone pairs, it has a steric number and a coordination number equal to 5 . According to VSEPR Theory, this corresponds to a trigonal bipyramidal molecular geometry.