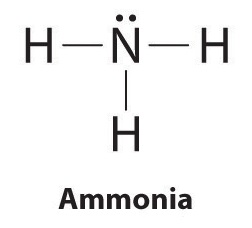
What is the steric number of #NH_3#?
1 Answer
Explanation:
For a given molecule, the steric number is calculated by counting how many regions of electron density surround the central atom.
A region of electron density can be
- a single, double, or triple bond -- all three count as one region of electron density
- a lone pair of electrons
In order to calculate the steric number of nitrogen, which is the central atom in ammonia,
The molecule will have a total of
The nitrogen atom will form single bonds with the three hydrogen atoms, with each of these bonds accounting for
The Lewis structure for ammonia will thus looks like this
Notice that the central atom is surrounded by a total of
- three single bonds
- one lone pair of electrons
You can thus say that the nitrogen atom has a steric number equal to
In this case, a steric number equal to