What is the strongest known acid?
1 Answer
Mar 29, 2018
Triflic acid is fairly dangerous, with
Fluoroantimonic acid is probably one of the (if not the) most acidic species, with
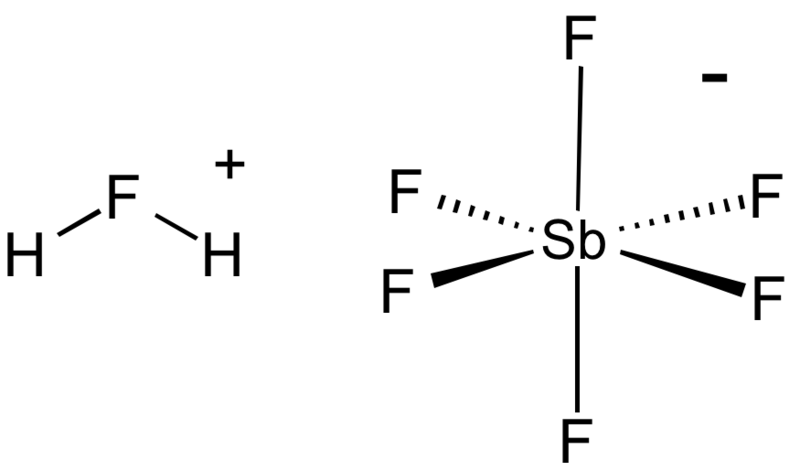
Those hydrogens on that fluorine are extra nasty, they'll protonate even hydrocarbons I'd imagine. For comparison, sulfuric acid is a typical strong acid, with


