What represents the area in an atom where an electron most likely will be found?
2 Answers
Orbitals.
Explanation:
An orbital is, more correctly, represented by a wave function or state function,
The probability density,
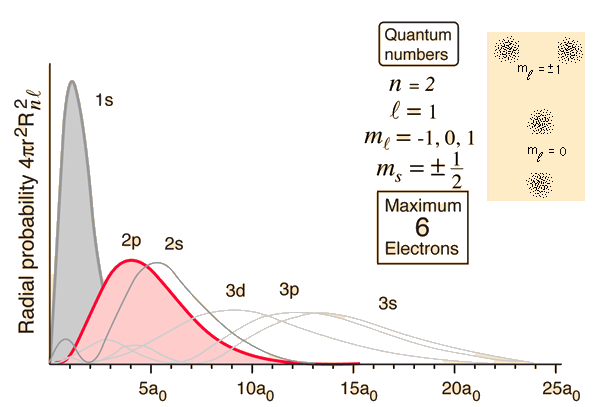
For example, the radial distance of the highest probability to find a
In organic chemistry one needs to be most familiar with the
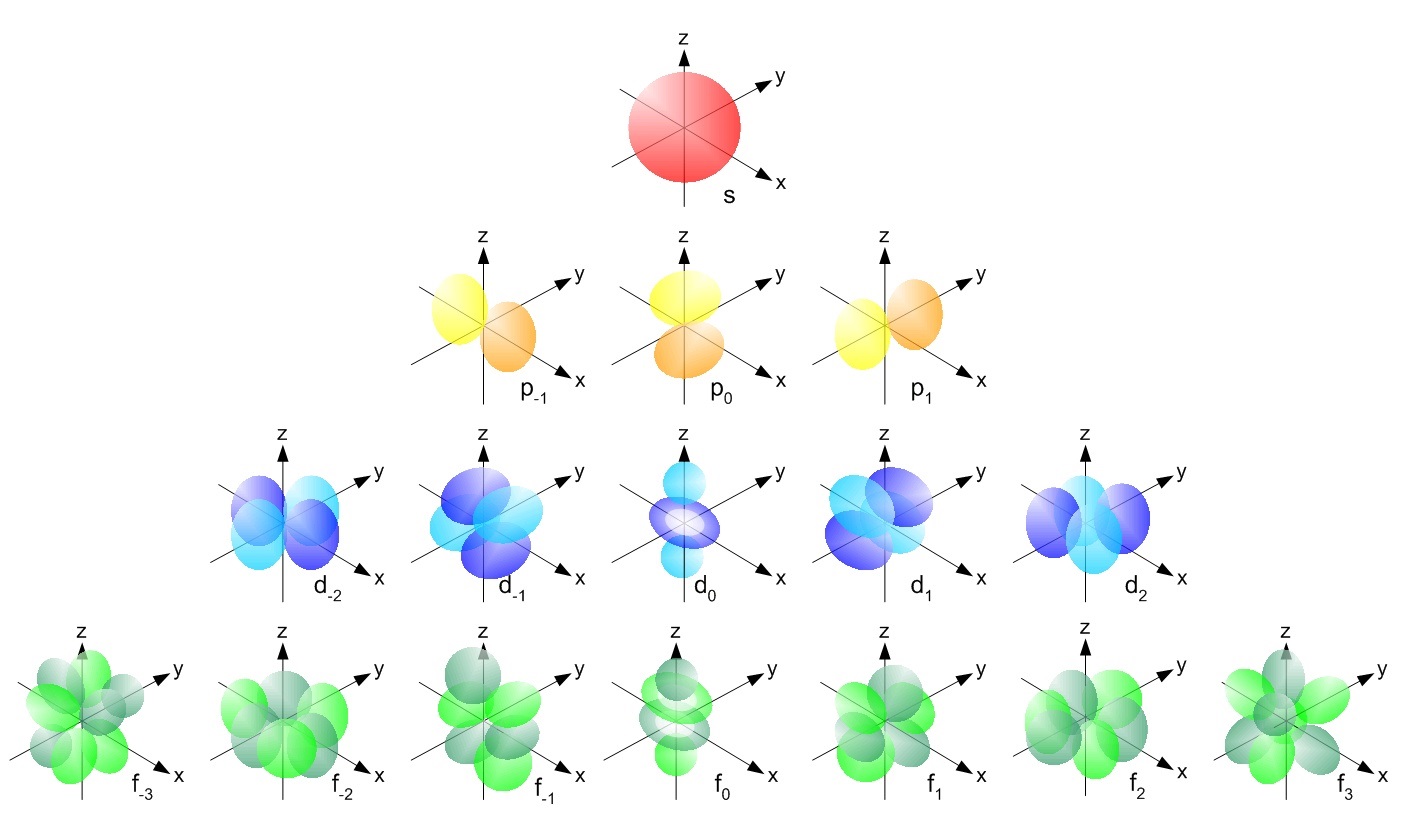
For example, the orbitals for the electrons in carbon, and the associated quantum numbers are given below. The nucleus of the carbon atom would reside at the center of the x,y,z coordinate.

Image => may be helpful...
Explanation:
Radial and Angular Nodes of Electronic Orbitals