Which Lewis electron-dot diagram is correct for a #"S"^(2-)# ion?
1 Answer
Jun 10, 2016
...
Explanation:
Sulfur belongs to group 16. It has 6 valence electrons. A
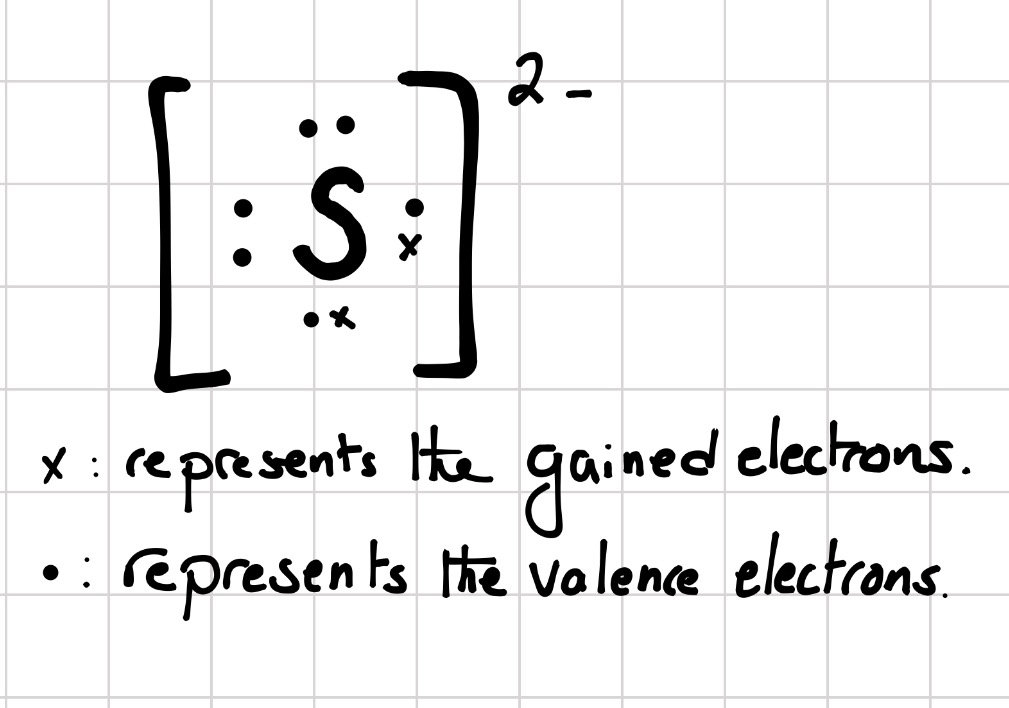
...
Sulfur belongs to group 16. It has 6 valence electrons. A

