Why is cohesion important in biology?
1 Answer
Cohesion is the property of a liquid to stay together. This is important in many parts of the biology, for example, the transport of water to all the leaves in a tree.
Explanation:
Cohesion is caused by interactions between the same sort of molecules. If we talk about adhesion, we mean the attractive interactions between different types of molecules.
It is easy to visualise cohesion since it is all around us! Just look at this picture below of a droplet sticking together instead of spreading out equally.

This effect is caused by interactions between the molecules. One type of these interactions is the hydrogen bond, formed between water. In the picture below these hydrogen bonds are depicted.
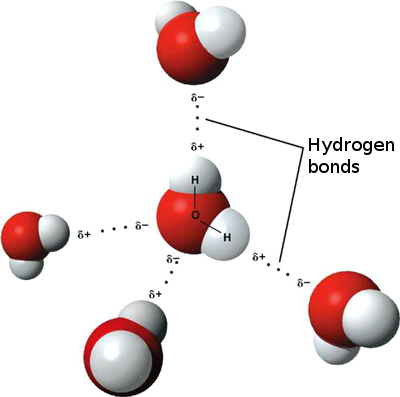
The positive and negative charges on these molecules attract each other, which is why the water droplet stays together.
Now, where do we find this in nature?
A nice example of this effect is found in trees! To transport the water up to the highest leaves, cohesion is one of the interactions that helps within this process, because it sticks together. Of course, not only cohesion is the cause of this, other interactions that help are adhesion, tree root pressure and the evaporation pressure on the leaves.
If these forces get an amount of water molecules a bit higher, other molecules are attracted to these molecules by cohesion and will easily follow. Therefore it cost less energy to transport all of them up!
If you put a water droplet on a table and you put a little stick into it and move it a bit around, you will find that (most) of the water will stick to your stick, while you do not even touch them with the stick! This is the same principle.
