Why is the relative stability of radicals different than that of carbocations?
1 Answer
Nov 16, 2014
The relative stability of radicals is different from that of carbocations because they have different numbers of valence electrons.
Free radicals have only 7 electrons in their valence shell. They are higher in energy than atoms with 8 valence electrons.
Carbocations are also electron-deficient species. Since carbocations have only 6 valence electrons, they are higher in energy than free radicals.
We know this, because many carbocations rearrange to become more stable. Free radicals seldom rearrange.
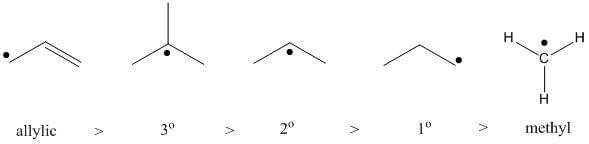
The same factors that stabilize carbocations stabilize free radicals.
The order of stability of carbocations is
The order of stability of free radicals is
Notice the difference? Neither do I.