Why is there an ionic radius trend?
1 Answer
Well, there is not much of a trend.... considering that metals are prone to form cations, and non-metals are prone to form anions...
Explanation:
We know that atomic size is a function of (i) atomic number
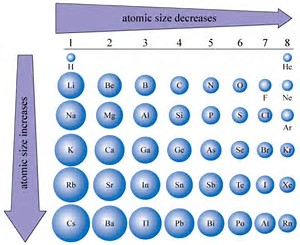
On the other hand, with respect to IONS, metals tend to LOSE electrons upon formation of ions, i.e. upon oxidation, and non-metals tend to gain electrons in a reductive process. The daughter cations, and anions should be respectively, MUCH SMALLER, and MUCH LARGER than the parent atom. Why? Well, reasonably, the atomic size is defined by the radius of the valence, the outermost electron, and the ionic radius of the CATION should be much reduced compared to the parent atom, and daughter anion formed by reduction should have a substantially greater radius.

Do these data support what I have argued? Units are in

