Absolute Zero
Key Questions
-
Answer:
It is the point at which particle motion stops for monatomic ideal gases. However, molecules will still vibrate.
Explanation:
All temperatures above absolute zero will cause particles in any material to move/vibrate slightly, as temperature gives particles Kinetic energy, according to the equipartition theorem for monatomic ideal gases:
#K_(avg)=3/2k_BT# #k_B# = Boltzmann's constant =#1.38065 times 10^-23 J//K#
#T# = absolute temperature (Kelvin)At absolute zero,
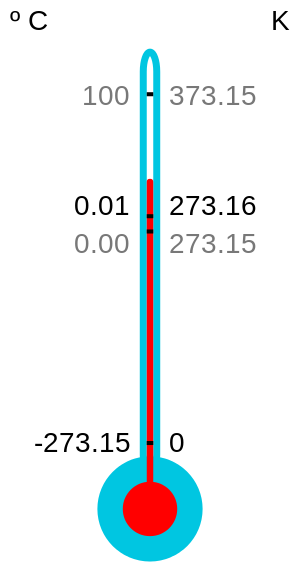
#T = "0 K"# , so there is effectively no average kinetic energy of the molecules. (although the state of absolute zero is more of a concept as it has not yet been achieved). This means that particle motion ceases in monatomic gases.Absolute zero is also the foundation for the Kelvin scale, as
#"0 K" = -273.15^@ "C"# is the coldest that you can ever hope to get. -
Absolute zero is the temperature at which enthalpy and entropy of ideal gas reaches zero.
In Kelvin scale this temperature is taken as
#0# Kelvin.