Naming Carboxylic Acid Derivatives
Key Questions
-
They have many derivatives, but some are esters, amides, anhydrides, and acid chlorides. I'm assuming you only mean the most similar-looking ones, so not nitrile (although acid-catalyzed hydrolysis of a nitrile gives a carboxylic acid).
The carboxylic acid I'll use is acetic/ethanoic acid.
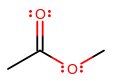
This ester here would be called methyl acetate or methyl ethanoate because a methyl group is attached to the non-carbonyl oxygen, and the original carboxylic acid was acet-ic or ethano-ic acid.
Retain the prefix "acet" or "ethano", replace "ic acid" with "ate" for "acet" or "oate" for "ethano", and place the name of the new substituent first.
e.g. sodium acetate, not acetate sodium (backwards), sodium acetoate (mixed up "ate" with "oate"), sodium ethanate (same as previous), nor sodium ethanolate (this is an alkoxide!).
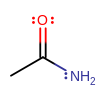
This amide here is just called acetamide or ethanamide because the original was called acet-ic or ethano-ic acid.
Retain the prefix, take out the suffix "ic" for acetic or "oic" for ethanoic, and add "amide".
Not aceticamide (although acetic acid amide is acceptable), ethanyl amide (why not ethyl like in many other naming cases?), nor ethanol amide (that is a real type of compound but completely different).
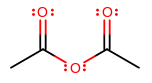
Again, the same pattern. This anhydride here is called acetic anhydride, or ethanoic anhydride. You might also say acetyl acetate or ethanoyl ethanoate to indicate that it is an ester ("ethanoate" or "acetate") attached to an acetyl/ethanoyl group---a two-carbon carbonyl group.
All you do is replace "acid" with "anhydride".
Not acetate anhydride (acetic!), ethanoate anhydride (ethanoic!), acetanhydride (two words, not one), acetyl anhydride (acetyl acetate!), nor ethanoyl anhydride (ethanoyl ethanoate!).
If the anhydride is asymmetrical, order the carboxyl substituents according to alphabetical order. e.g. methanoic ethanoic anhydride or acetic formic anhydride for the same molecule could be used, etc (it doesn't matter which carboxyl substituent is on which side because you can flip the molecule).

Lastly, this acyl/acid chloride here is called acetyl chloride or ethanoyl chloride. You can see another pattern here.
Retain "acet" or "ethano", and replace "ic acid" with "yl halide".
Not acyl chloride---unless it is specifically acetyl chloride (that's the general name for the family otherwise), acetic chloride (acetyl!), nor ethanoic chloride (ethanoyl!).
-
Answer:
Primary amides are named by replacing the –ioc acid ending of the carboxylic acid with the ending -amide.
Explanation:
You can view a 1° amide as if it were prepared by reacting a carboxylic acid with ammonia.
A typical equation might be
#underbrace("CH"_3"COOH")_color(red)("ethanoic acid") + underbrace("NH"_3)_color(red)("ammonia") → underbrace("RCONH"_2)_color(red)("ethanamide") + "H"_2"O"# To name the product, you simply replace the -oic acid ending of ethanoic acid with the ending -amide.