Introduction to IUPAC Nomenclature
Key Questions
-
Identify the longest carbon chain. (This chain is called the parent chain.)
For example, in 2,4,6,8-tetramethyl-2,4-hexadiene below, the parent chain is highlighted:
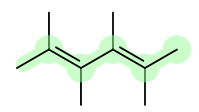
Identify all of the substituents (groups of atoms branching out from the parent chain of choice)
The substituents in the above structure would be the methyl groups.
Number the carbons of the parent chain from the end that gives the substituents the lowest numbers possible. This is significant usually when the molecule is asymmetrical.
- This means that 1,2,4-trimethylpentane is preferred over 2,4,5-trimethylpentane, for example, because the first number has 1 < 2.
- If two or more side chains are in equivalent positions, assign the numbers in ascending order. So, 1,2,4, not 1,4,2 or 4,1,2, etc.
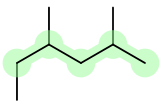
If the same substituent occurs more than once, the location of each point on which the substituent occurs is given. In addition, the number of times the substituent group occurs is indicated by a prefix (di, tri, tetra, etc.).
So, in 1,2,4-trimethylpentane (or 2,4-dimethylhexane), there are three (tri for three) methyl groups branching out from carbons 1, 2, and 4.
If there are two or more different substituents, they are listed in alphabetical order using the base name (ignore the prefixes).
Exceptions where the prefix is involved include:
- iso, if it is one word with the other substituent(s); otherwise, ignore the prefix. For example, either isobutylmethyl or methyl isobutyl are fine.
- sec- and tert- are not alphabetically significant unless compared with each other.
For example, you can say isobutylmethyl ketone, OR methyl isobutyl ketone.
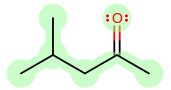
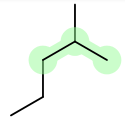
An example of a normal case would be 1-ethyl-2-methylpropane, where methyl comes after ethyl.
If chains of equal length are competing for selection as the parent chain, then the choice goes according to this hierachy:
1) the chain which has the greatest number of side chains.
2) the chain whose substituents have the lowest position numbers.
3) the chain having the greatest number of carbon atoms in the smaller side chain.
4) the chain having the least branched side chains.Example:
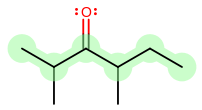 vs.
vs.
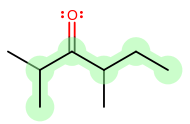
Number 1 is irrelevant because both have three substituents.
Number 2 is irrelevant because both have positions 2, 3, and 4 occupied.
Number 3 is irrelevant because the smallest sidechains are identical.
Number 4 is irrelevant because none of the sidechains are branched.A cyclic (ring) hydrocarbon is designated by the prefix cyclo- which appears directly in front of the base name.
An example is 1-hydroxy-2-methoxycyclohexane, where hexane is the base name, an
#"OH"# , a hydroxyl group, is on carbon 1, and a#"CH"_3"O"# , a methoxy group, is on carbon 2.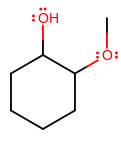
The parent chain is numbered so that the multiple bonds have the lowest numbers if there is only one type of multiple bond (double and triple bonds have priority over alkyl and halo substituents).
Example:
3-bromo-4-fluoro-1-butene, or 3-bromo-4-fluorobut-1-ene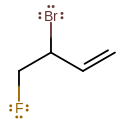
When both double and triple bonds are present, numbers as low as possible are given to double and triple bonds even though this may at times give "-yne" a lower number than "-ene". When there is a choice in numbering, the double bonds are given the lowest numbers.
When both double and triple bonds are present, the -en suffix follows the parent chain directly and the -yne suffix follows the -en suffix (notice that the e is left off, -en instead of -ene). The location of the double bond(s) is(are) indicated before the parent name as before, and the location of the triple bond(s) is(are) indicated between the -en and -yne suffixes.
Example: prop-1-en-3-yne
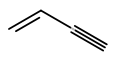
For a branched unsaturated acyclic hydrocarbon, the parent chain is the longest carbon chain that contains the maximum number of double and triple bonds.
If there are two or more chains competing for selection as the parent chain (chain with the most multiple bonds), the choice goes to (1) the chain with the greatest number of carbon atoms, (2) the # of carbon atoms being equal, the chain containing the maximum number of double bonds.
For example:
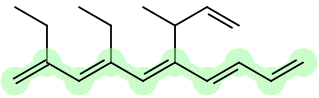
You could call this 2,4-diethyl-6-methylallyl-1,3,5,7,9-decapentaene.
If there is a choice in numbering not previously covered, the parent chain is numbered to give the substituents the lowest number at the first point of difference.
-
IUPAC names aim to give each chemical a clear, unique, and self-explanatory indicative name so that anyone can tell what the chemical is from its name.
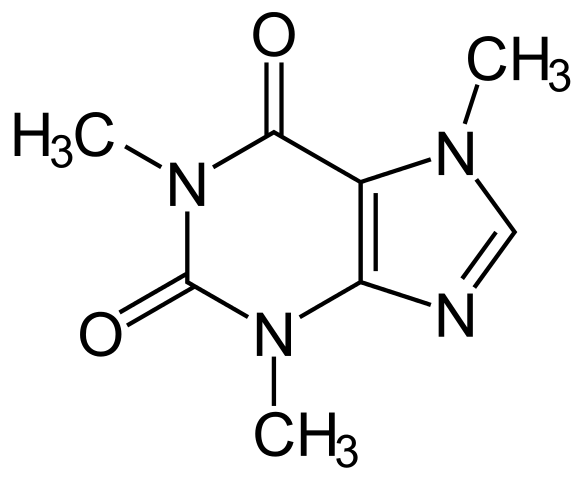
A nice example is "Caffeine", where its IUPAC name is 1,3,7-Trimethylpurine-2,6-dione. For chemists familiar with IUPAC naming rules can easily relate that IUPAC name with its structure as shown in the picture below.
-
IUPAC is the International Union of Pure and Applied Chemistry. They decide the chemical nomenclature of newly discovered elements or compounds. It is based in Zurich, Switzerland, and was established in 1919 as the successor of the International Congress of Applied Chemistry for the advancement of chemistry.
The organization is best known for its works in standardizing nucleotide base sequence code names; publishing books for environmental scientists, chemists, and physicists; and leading the way in improving education in science. Its current president is Kazuyuki Tatsumi, and has been run by many different committees with different responsibilities.