Question #ae740
1 Answer
Here's how you can think about the formation of the hydrogen molecule from the VBT perspective.
Explanation:
I'm going to focus on the idea behind how Valence Bond Theory (VBT) accounts for the formation of the hydrogen molecule,
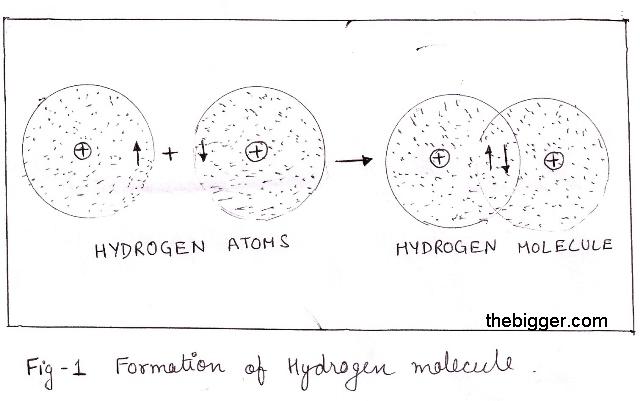
The main idea behind the VBT is that the chemical bonding that exists between two atoms is caused by the overlapping of partial-filled atomic orbitals.
In your case, the overalpping of the 1s-orbital of one hydrogen atom and the 1s-orbital of another hydrogen atom will result in the formation of a single bond, or sigma bond.
This overlapping between the two half-filled orbitals will produce a region of higher electron density.
The two electrons that each orbital contributes, which are called bonding electrons, will now be located directly between the two nuclei.

So, here's what's actually happening when two hydrogen atoms bond. As two hydrogen atoms approach each other, the attraction forces that exist between the nuclei and the electrons will overpower the repulsive forces that exist between the two electrons and between the two nuclei.
This causes the atoms to continue to approach each other until a sweet spot is reached where the attractive forces are perfeclty balanced by the repulsive forces.
This is where the potential energy of the system is at its lowest point.
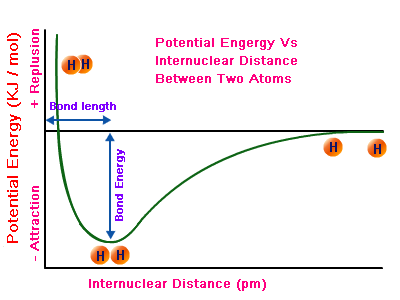
This "perfect distance" between the two atoms is called the bond length.
When the two atoms are at bond length, their orbitals overlap, or interpenetrate. This is where the maximum probability of finding the two bonding electrons, which at this point have opposite spins with respect to each other, will be.
The overlapping of two s-orbitals will produce a s-s sigma bond.