What is the geometry and hybridization of #"AuCl"_4^-#?
1 Answer
Warning! Long Answer. The
Explanation:
CRYSTAL FIELD THEORY
Crystal field theory postulates that
#"d"# orbitals pointing directly at an axis are most destabilized by electrostatic interactions with a ligand.#"d"# orbitals pointing away from an axis are least destabilized by electrostatic interactions with a ligand.

(From www.slideshare.net)
For example, in an octahedral field, the diagram above shows that:
- the
#"d"_(z_2)# and#"d"_(x^2-y^2)# orbitals are most destabilized. - the
#"d"_text(xy), "d"_text(xz)# , and#"d"_text(yz)# orbitals are least destabilized.
In a square planar complex, the four ligands are only in the
The absence of ligands along the
When you work it out, there turns out to be four different energy levels in a square planar field:

The
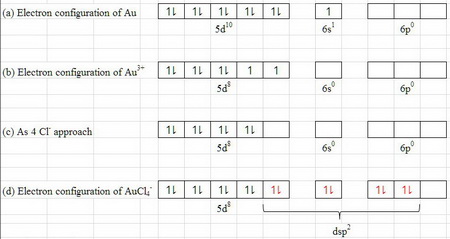
(a) The electron configuration of
(b) The electron configuration of
(c) As the 4
(d) The
These orbitals can accept a lone pair from each of the chloride ions and form
The bonds point to the corners of a square, forming a square planar molecular geometry.
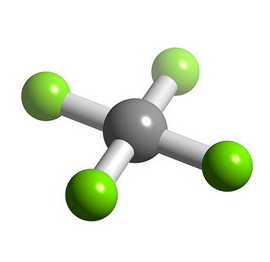
(From www.chemtube3d.com)