Question #0442e
1 Answer
Feb 4, 2018
Na is bigger than Mg because of one less proton in the nucleus to attract the outermost electron cloud
Explanation:
See the image below
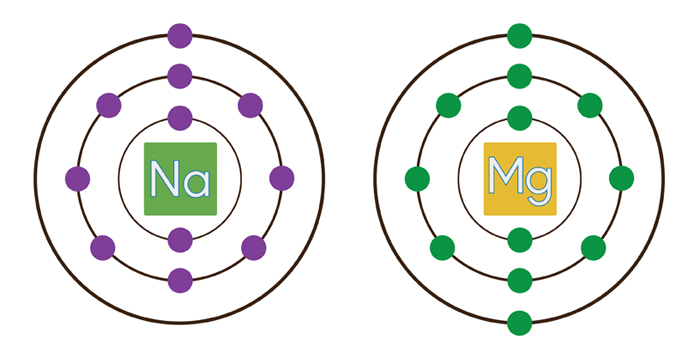 www.learner.org/courses/chemistry/images/lrg_img/Mg_NaElectronShellsjpg
www.learner.org/courses/chemistry/images/lrg_img/Mg_NaElectronShellsjpg

