How many electrons are in the highest occupied energy level of a group 5A element?
1 Answer
Explanation:
The thing to remember about a main-group element is that the group number tells you the number of valence electrons said element has.
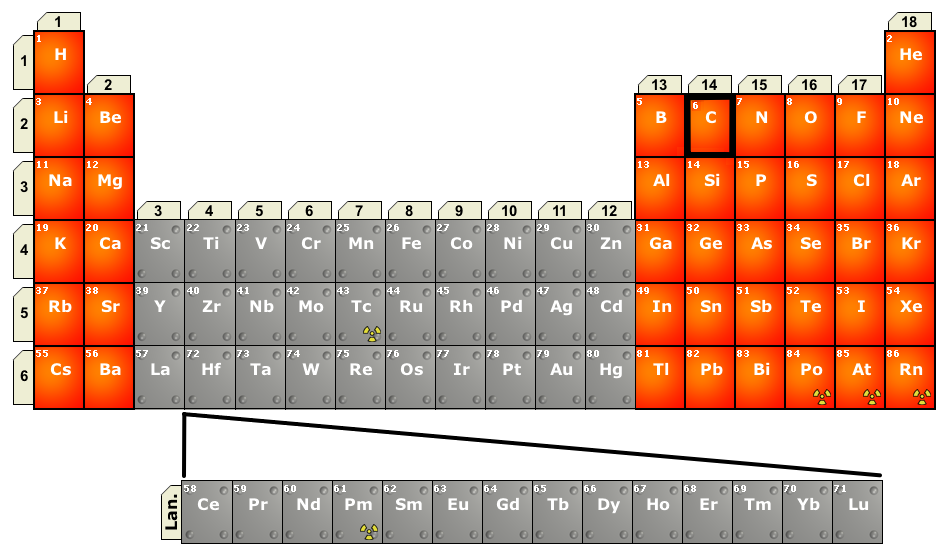
Main-group elements are located in groups
You thus have
"Group 1 " -> " Group 1A" "Group 2 " -> " Group 2A" "Group 13 " -> " Group 3A" "Group 14 " -> " Group 4A" "Group 15 " -> " Group 5A"
vdots
and so on.
Now, your element is said to be located in group
"Group"color(white)(a)color(red)(5)"A" -> color(red)(5)color(white)(a)"valence electrons"
"Group 1"color(red)(5) -> color(red)(5)color(white)(a)"valence electrons"
As you know, an element's valence electrons are simply the electrons located on the outermost energy level, i.e. on the highest occupied energy level.
Therefore, you can say that an element located in group

