What is an acid catalyzed hydro-alkoxy addition?
1 Answer
Dec 13, 2014
An acid catalyzed hydro-alkoxy addition is the addition of an alcohol to a C=C double bond to form an ether.
An example is the addition of methanol to 2-methylpropene to form t-butyl methyl ether.

This is an electrophilic addition reaction.
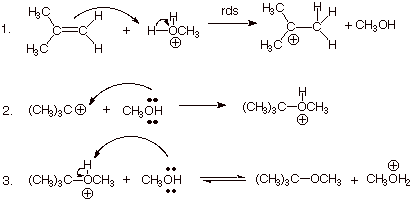
In Step 1, a hydronium or oxonium ion is attacked by the π bond.
In Step 2, the alcohol attacks the carbocation and forms an oxonium ion.
In Step 3, the alcohol deprotonates the oxonium ion to form the ether.
Here’s a video on the addition of alcohols to alkenes.

