What is the Lewis structure for #CN_2H_2#?
1 Answer
In order to determine the Lewis structure of methanediimine, or
This is done by adding the valence electrons of each of the atoms that form the molecule
Carbon contributes 4 valence electrons, each of the nitrogen atom acontributes 5, and each of the hydrogen atoms contributes 1 valence electron.
So, start by connecting all the atoms with single bonds.
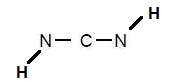
Each single bond has 2 electrons, so we've used up
Now you only have to work with
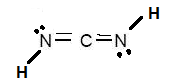
This will complete the Lewis structure of the molecule, since all

