What undergoes sn1 reactions?
1 Answer
Jun 14, 2018
Tertiary halogenoalkanes.
Explanation:
-
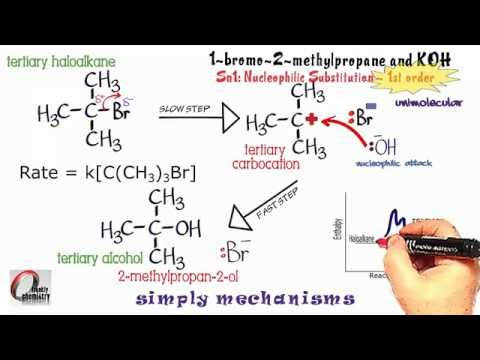
Here, the first slow step involves only the heterolytic fission of the C-X bond (where X is the halogen).
-
As a result of this fission, you get a tertiary carbocation and
#X^-# . -
It then follows that the nucleophile (like
#OH^-# or#CN^-# ), attacks the carbocation quickly.
This produces the products.
An example involves the reaction of 2-bromo methylpropane with NaOH in water. You get as a result methyl proapan-2-ol and NaBr.
In

