Why are electrophilic addition reactions the characteristic reactions of alkenes?
1 Answer
Apr 5, 2018
Here's an electrostatic potential map for ethylene,
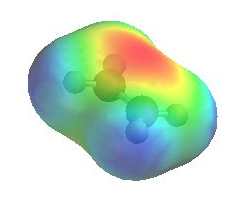
Where red indicates high electron density.
In ionic reactions, what has the most electron density?
A nucleophile.
Hence, addition reactions of myriad of materials (an electrophilic addition occurs first in most mechanisms) about the planar double bond with different regioselectivity and stereospecificity are the most common reactions.
Here's a non-exhaustive list of a few ways we can leverage


