Why is the polarizability of an anion directly proportional to its size?
1 Answer
Because larger anions have larger electron clouds that are easier to distort.
Explanation:
As you know, an anion's size is determined by how far away from the nucleus its outermost shell is.
As you move down a group of the periodict table, the atomic size increases because the outermost electrons are being added further and further away from the nucleus.
This carries over to ionic size as well. In addition to the fact that these outermost electrons are further away from the nucleus, they are also increasingly better screened from the nucleus by the core electrons.
This means that the attraction between these outermost electrons and the nucleus is not as significant as it is for the electrons located on lower energy levels.
Polarizability represents the ability of an anion to become polarized. In order for an anion to become polarized, its electron cloud must be distorted.
This implies that the easier it is for an anion's electron cloud to be distorted, the more polarizable that anions is.
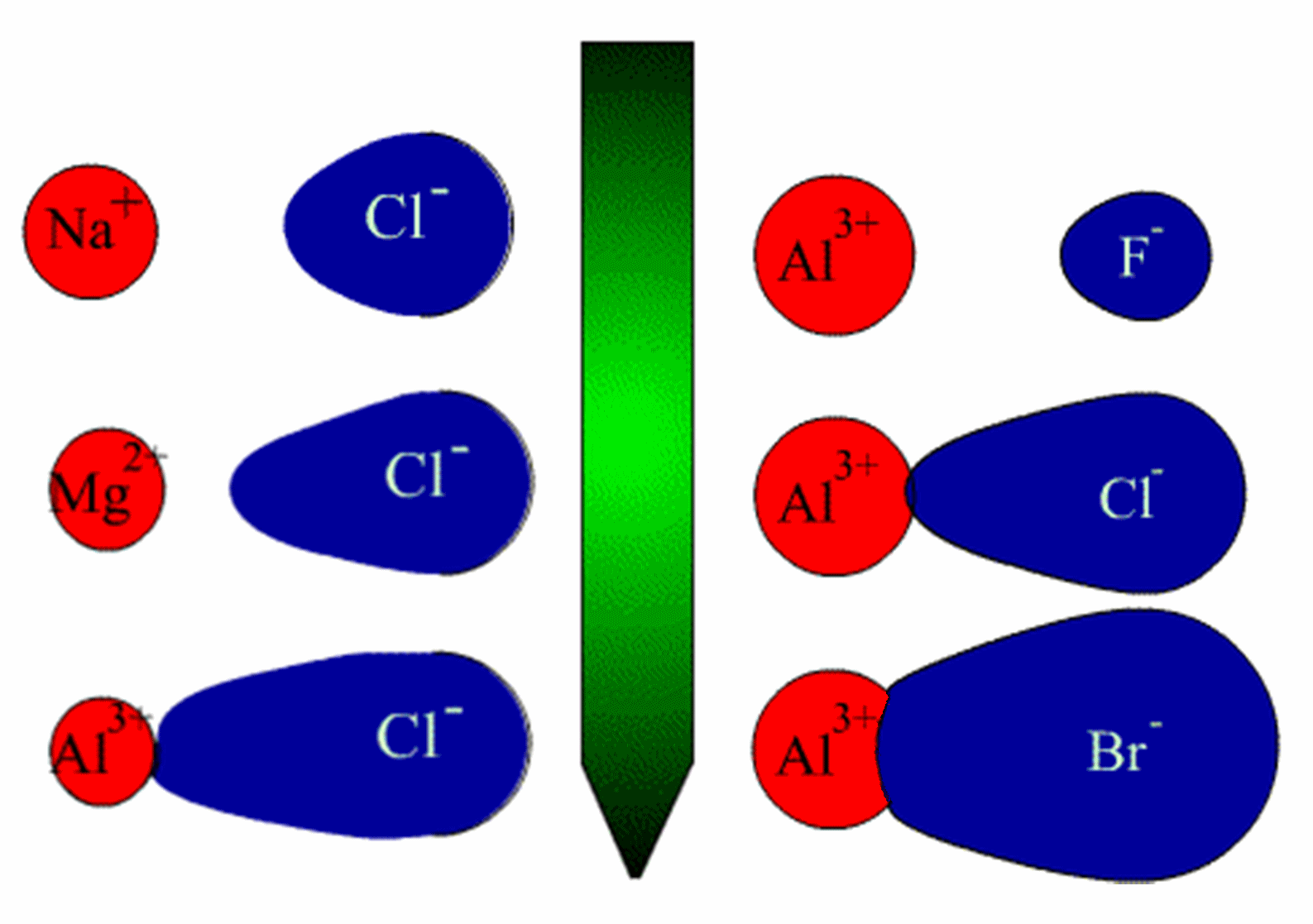
This is why ionic size is directly proportional to anion polarizability. The larger the anions are, the more loosely held its outermost electrons are, since they are located further away and are better screened from the nucleus.
This ultimately means that the electron clouds are very easy to distort, which is why larger anions are more polarizable than smaller anions.
So, when the positively charged cation comes close to a larger anion, like iodide,
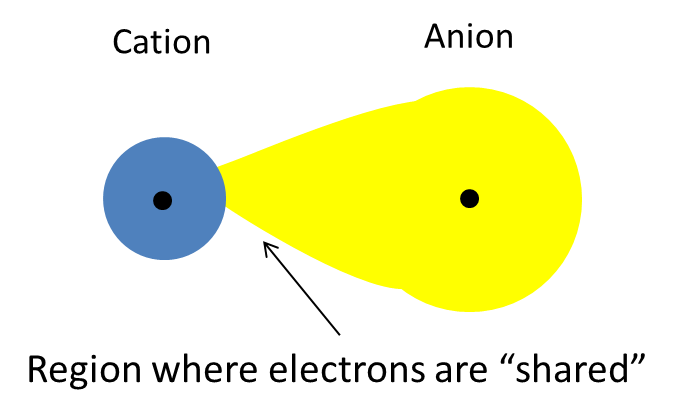
By comparison, fluoride's outermost electrons located very close to the nucleus and don't benefit from any significant screening, which is why fluoride's electronc cloud is very compact and not very easy to distort.