Question #2726a
1 Answer
Jul 24, 2015
Because it reduces steric strain, i.e. it gives the substituents more space for their orbitals and minimizes repulsive forces. Minimizing these repulsive forces between electrons minimizes the energy of the molecule, and molecules always want to have as low-lying of an energy state as possible.
Here are three depictions of the interactions between adjacent bonding electron pairs:
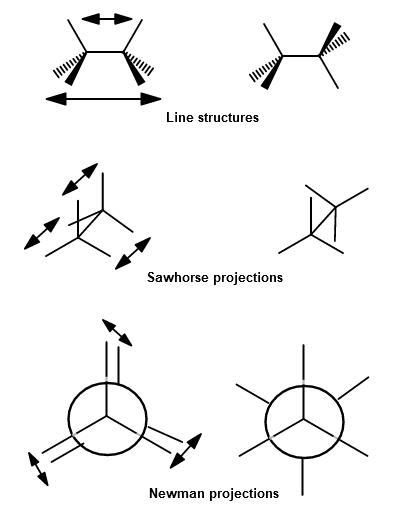
This gives a barrier to
(For reference,

