What is the most stable conformer for 3,3-dimethylhexane, viewed along the #C_3-C_4# bond using Newman projections?
1 Answer
The most stable conformer for 3,3-dimethylhexane viewed along the
Start by drawing the bond line notation for the compound
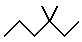
Notice that you have one ethyl group and two methyl groups attached to
Start by using a Newman projection template to draw the first staggered projection
Projection 1
Now rotate the front carbon by
Projection 2 and Projection 3
Projection 2 and projection 3 will be less stable than projection 1 because in both cases you have gauche interactions between two bulky ethyl groups and a methyl group.
By comparison, projection 1 has the two ethyl groups in anti position, the repulsion between two methyl groups and one ethyl group being a little less unstable than the repulsion the other two staggered conformers have.
Therefore, projection 1 describes the most stable conformer for 3,3-dimethylhexane viewed along the

