What is the order of increasing #"S"_text(N)2# activity of the following compounds?
#"CH"_3"CH"_2"X"#
#"PhCH"_2"X"#
#("CH"_3)_2"CHX"#
#"CH"_2"=CHCH"_2"X"#
1 Answer
Explanation:
The
(Adapted from SlideShare)
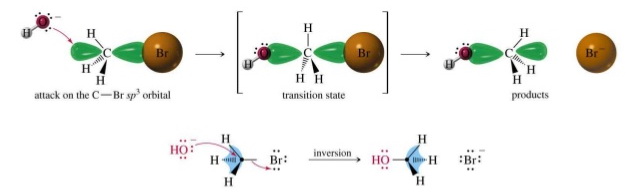
The rate of the reaction depends on steric and mesomeric effects.
The mesomeric effect causes allylic and benzylic halides to have faster
This is because the π-electrons can overlap with the partially broken and formed bonds in the transition state.
This lowers the activation energy and increases the reaction rate.
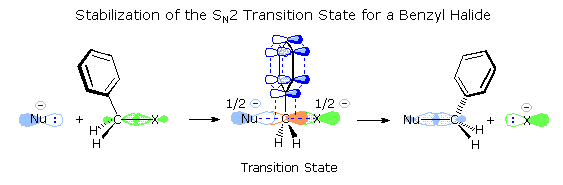
The phenyl group can delocalize the partial charge better than a vinyl group can.
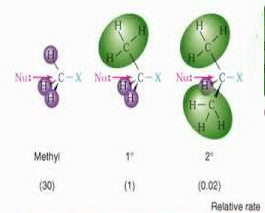
The extra methyl group in isopropyl halides (on the right) makes it more sterically hindered.
(Adapted from YouTube)
Thus, the order is

