Question #47fbe
1 Answer
Jan 2, 2018
Here's what I get.
Explanation:
(a) 1-Chloro-3-methylcyclopentane with aqueous
This is an
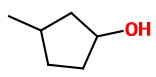
(b) 2-Bromo-2-methylpentane with ethanolic
This is an
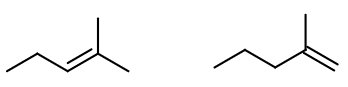
(c) Mechanism
The hydroxide ion reacts with ethanol to produce ethoxide ion in an equilibrium reaction.
The ethoxide ion is a stronger base than
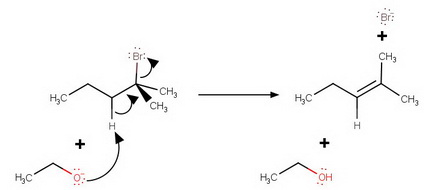
(d) "We leave it as an exercise for the student" to compare and contrast the

