Question #382fd
1 Answer
Jul 24, 2017
Proline gives a yellow colour because it is a secondary amine.
Most amino acids are primary amines with the general structure
Except for proline and hydroxyproline, all the α-amino acids are oxidized by ninhydrin to give the same intensely colored purple anion.
The central
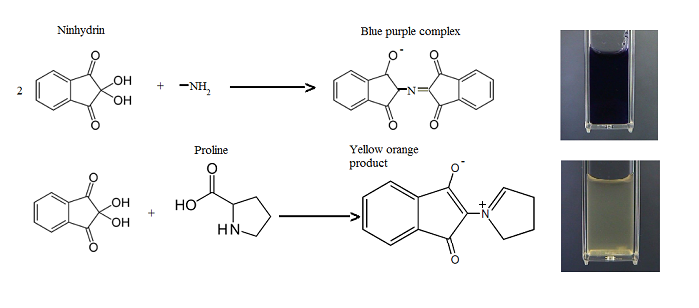
(Adapted from Biochemistry is a good thing - WordPress.com
Proline reacts differently because it is a secondary amine.
The product is a yellow-coloured zwitterion.

