Question #115a7
1 Answer
Apr 11, 2017
It is a nucleophilic substitution
Explanation:
As you know, KCN will dissociate into
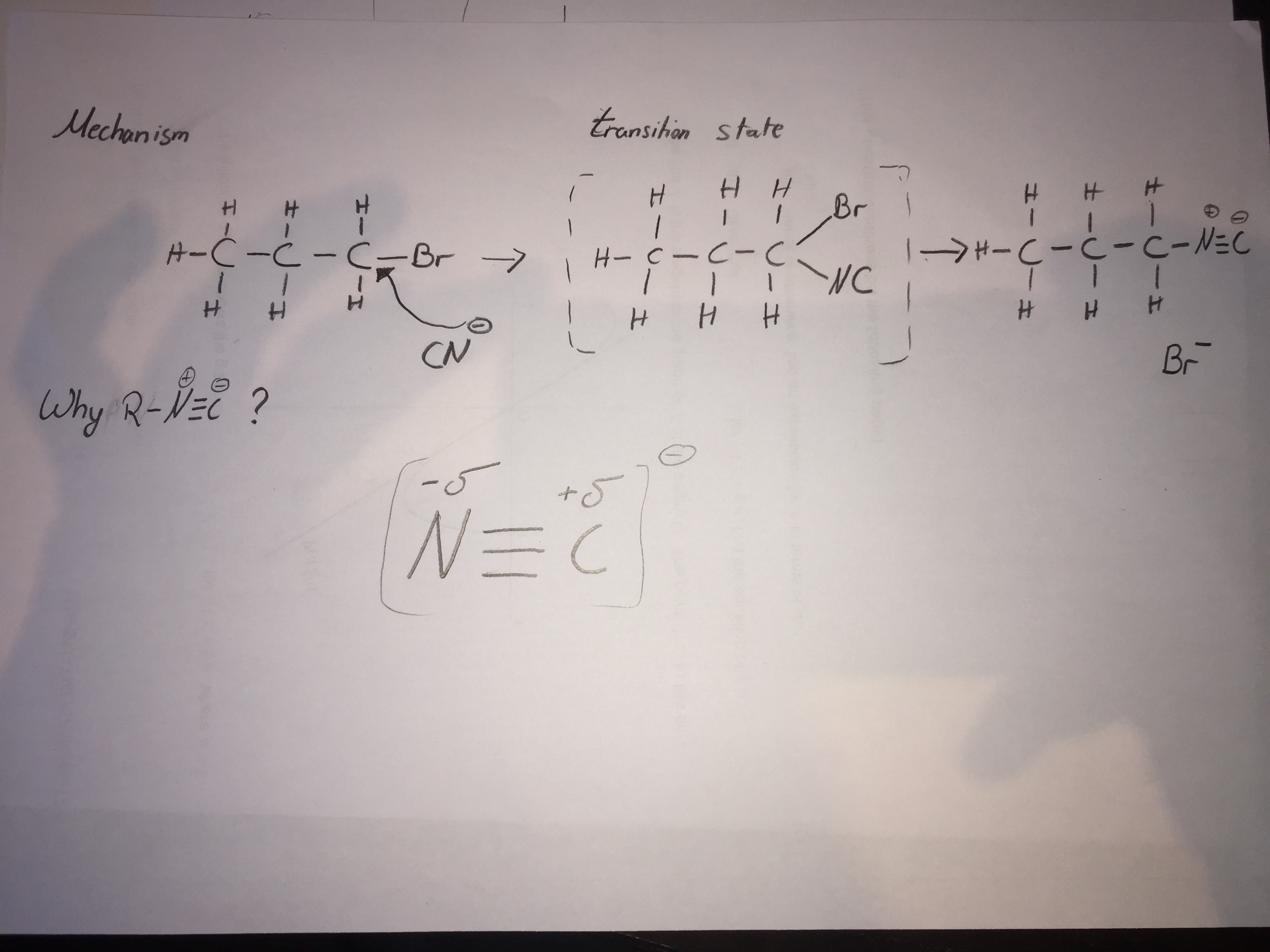
It is a nucleophilic substitution
As you know, KCN will dissociate into
