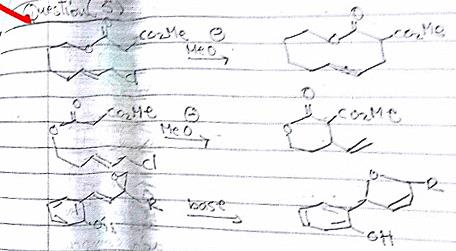
Can anyone help me with these cyclization questions?
2 Answers
Here are my suggestions.
Explanation:
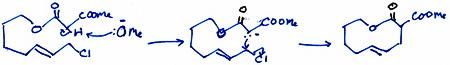
(a) Malonate
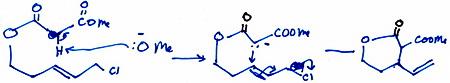
(b) Malonate Conjugate
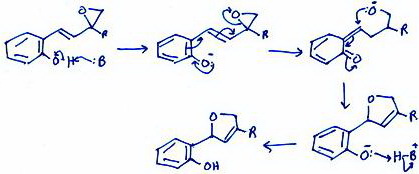
(c) Epoxide Ring Opening
Question 1: probably the easiest, this is standard carbanion and enolate chemistry.
Explanation:
The substrate is:
This is the enolate, and this is the carbanion:
This reacts as the carbanion to effect ring closure at the allylic carbon on the other end of the chain to eliminate halide.
The way it is drawn on the paper is much more intuitive than the way I have drawn it in this editor, because you can see the potential
The point is that


