Consider Diels-Alder reaction between two molecules of methylvinylketone.What product will be formed?
1 Answer
The Diels-Alder reagents will try to align in a benzene-like orientation to form a stable transition state.
Furthermore, the diene must be in an s-cis conformation to allow for favorable orbital overlap. Thus, the mechanism should look like this:
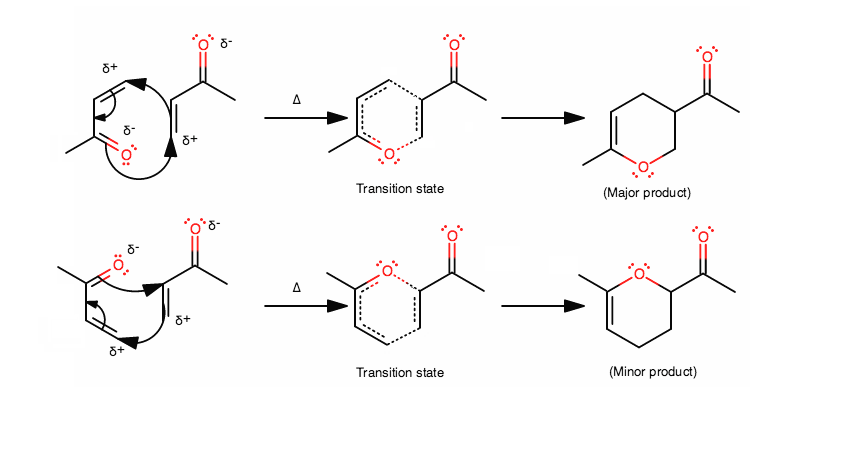
The positive end of the molecule lines up with the negative end of the molecule, so you should recognize that one of them have to be flipped vertically to form the major product.
If you don't see why this is, try drawing resonance structures of the molecule, donating the carbon
In addition, the carbon is the atom that accepts electrons here in its antibonding orbital, not the oxygen; the oxygen is already withdrawing electrons, so it is more nucleophilic than electrophilic.

