Write equations to show how 2,3-dimethylbutane may be prepared from each of the following compounds. (i)an alkene (ii)A grignard reagent (iii)a haloalkane (iv)a sodium alkanoate?
1 Answer
Aug 26, 2015
i) Starting from any alkene
You can create this from 2-butene, or an alkene with a double bond on carbon-2. It theoretically doesn't matter whether it's cis or trans.
I would do this:
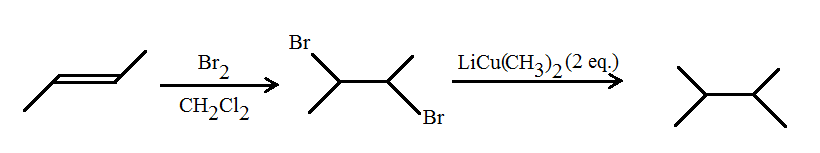
- Basic bromination in dichloromethane
- Add two equivalents of
#"LiCu"("CH"_3)_2# , a type of Gilman reagent, to essentially substitute both bromide groups with methyl groups like so
ii) Starting from a grignard reagent
I don't really see the point of starting from a Grignard reagent since it's usually a nucleophile... but:
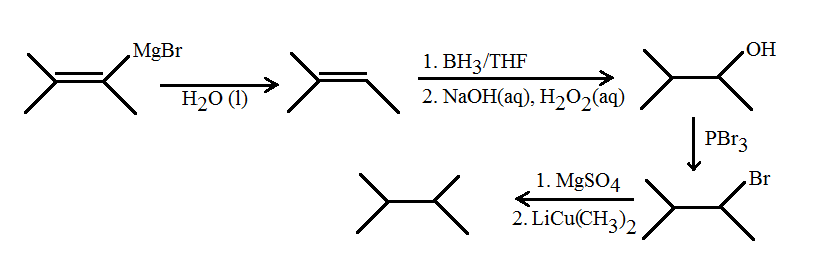
- Water gets rid of the magnesium bromide substituent and substitutes it with a hydrogen
- Hydroboration adds anti-Markovnikov to give a hydroxide on the carbon where the magnesium bromide once was
#"PBr"_3# substitutes the hydroxide with a bromide group#"MgSO"_4# acts as a drying agent to clear the reaction vessel of any water remaining from steps 1 and 2 (you may have used this in lab already); safe way of minimizing potential reactions with water#"LiCu"("CH"_3)_2# substitutes a methyl group in place of the bromide group
iii) Start from step 2 of part i) and do the same thing from that point on
iv) Starting from any sodium alkanoate
This'll take a while to do in real life...
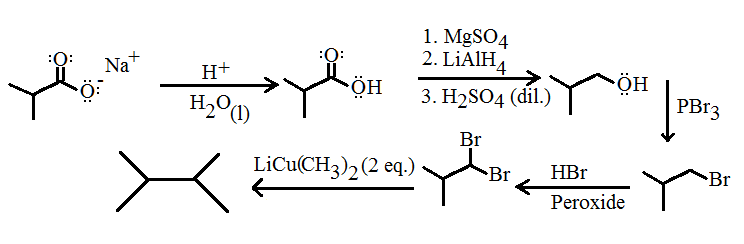
- Strong acid protonates the alkanoate to make a carboxylic acid
#"MgSO"_4# dries out the reaction vessel to prevent overly violent reaction in step 3#"LiAlH"_4# acts as a strong reducing agent that is capable of reducing a carboxylic acid down to the corresponding alcohol- Dilute sulfuric acid terminates the reducing process
#"PBr"_3# substitutes the hydroxide with a bromide group#"HBr"# with a peroxide causes a radical reaction; essentially, anti-Markovnikov addition of a proton to the dimethylated carbon (bottom left) and a bromide to the upper right carbon.- Two equivalents of
#"LiCu"("CH"_3)_2# substitutes with each bromide a methyl group (this is not likely to give that great of a yield due to the steric hindrance, but it's theoretical so it's OK)

