What is the energy profile in *cis*- and *trans*-3-bromo-6-methylcyclohexene?
1 Answer
The structure of 3-bromo-6-methylcyclohexane is
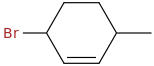 opsin.ch.cam.ac.uk
opsin.ch.cam.ac.uk
C-1 and C-4 are chiral centres, and there are no planes of symmetry, so there are
They exist as enantiomeric pairs of cis and trans isomers.
cis-3-bromo-6-methylcyclohexene
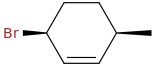 opsin.ch.cam.ac.uk
opsin.ch.cam.ac.uk
and
trans-3-bromo-6-methylcyclohexene
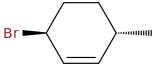 opsin.ch.cam.ac.uk
opsin.ch.cam.ac.uk
Cyclohexene exists in a half-chair conformation.
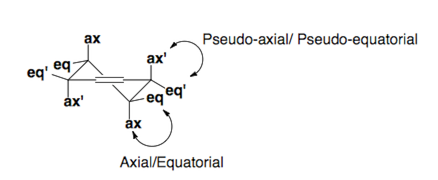 chemwiki.ucdavis.edu
chemwiki.ucdavis.edu
C6, C1, C2, and C3 are all in one plane. One carbon is above, and one carbon is below the plane. The substituents at C-3 and C-6 are pseudoaxial and pseudoequatorial.
The conformational strain is usually about half that in cyclohexanes, because a substituent has only one 1,3-diaxial interaction.
So, we expect the 1,3-diaxial CH₃ interactions to be about 3.8 kJ/mol. The 1,3-diaxial Br interactions should be about 1.4 kJ/mol.
In a cis-3,6-disubstutited cyclohexane, one substituent is always equatorial, and the other is always axial.
In the trans isomer, both substituents are either both equatorial or both axial.
trans-1,4-Dibromocyclohexane can exist in the diequatorial form with no 1,3-diaxial interactions. That is the most stable conformer.
The diaxial conformer would have (3.8 + 1.4) kJ/mol = 5.2 kJ/mol of steric strain. This is the least stable conformer
In cis-1-bromo-4-methylcyclohexane, the conformer with an axial CH₃ has 3.8 kJ/mol of strain. The conformer with an axial Br has 1.4 kJ/mol of strain.
In a cis-1,4-disubstutited cyclohexane, one substituent is always equatorial, and the other is always axial.
The major conformer will have the CH₃ group equatorial.

