Explain the mechanism of the Tishchenko reaction?
1 Answer
Interesting mechanism!
THE OVERALL TISHCHENKO REACTION
The overall reaction is an alcohol reacting with sodium metal at a low temperature (to minimize side reactions), forming an tri-oxo compound of sorts, which cascades downwards to form an ester.
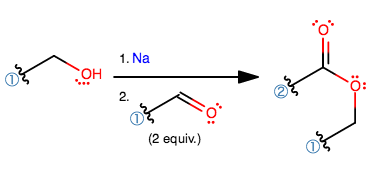
THE TISHCHENKO MECHANISM
Here's how I understood the mechanism.
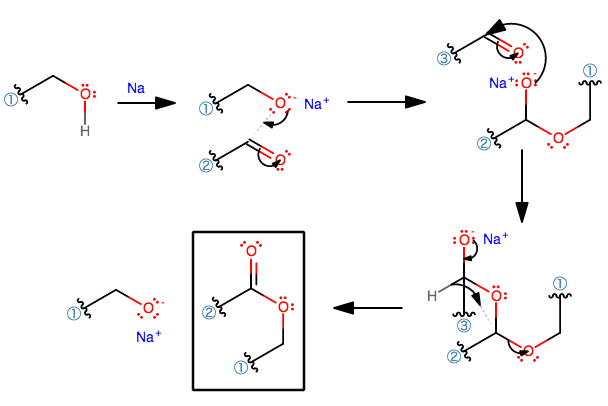
- Likely that this is a simple single-replacement:
#2"ROH"(l) + 2"Na"(s) -> 2"RONa" + "H"_2(g)# This generates the active nucleophile. - Then the aldehyde is placed into solution as the electrophile in the presence of the alkoxide nucleophile. The alkoxide attacks the aldehyde like it would a typical carbonyl compound.
- This repeats one more time to generate a tri-oxo compound.
- This is the interesting part. The rest, you've probably seen before if you've gotten to carbonyl chemistry in organic chemistry.
What happens here is known as a 1,3-hydride shift, which is a type of sigmatropic rearrangement. This is actually quite rare!
- The
#"C"-"O"^(-)# bond rotates to align the#p# orbital of oxygen with the#p# orbital of the electrophilic carbon (next to the (3) alkyl chain), then#pi# -donates to form a#pi# bond. - The hydride can then shift downwards to the carbon next to the (2) alkyl chain, bumping off the alkoxide.
The transition state likely looks like this:
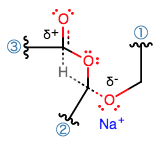
which is quite unusual (normally I would expect to see this kind of agostic hydrogen, across two atoms), but that is what the mechanism suggests.
The middle oxygen cannot accept electrons from the hydride (it is electron-donating, and has no compatible antibonding orbitals).
Instead, the hydride can
To maintain a standard octet valence, that carbon must donate electrons into the bottom oxygen's antibonding (
FINAL PRODUCT
The 1,3-hydride shift generates the final product, which is an ester consisting of an
The aldehyde that was bound with the (3)
SIDENOTE: Interestingly enough, one might expect that the middle
Well, if it did occur, then we would go backwards and reform the previous intermediate. So, perhaps there is a large excess of the alkoxide that the equilibrium is pushed towards formation of product.

