Chiral and Achiral Molecules
Key Questions
-
Because organisms may react differently to stereo-isomers.
Whenever there are asymmetric atoms in a molecule, there is the possibility of stereo-isomers. The simplest form are mirror images of each other, like a pair of gloves.
Most organisms are 'keyed' to processing only one of these forms, and that is the form that usually occurs as the only one in nature, because they are also produced by (other) organisms.
Chemical synthesis of compounds very often produces an mixture of both forms, called a racemic mixture, where only one form is effective, and the other form is not used or may even do damage.
One example of a chiral molecule is glucose, that naturally only occurs in the so-called right-hand variety, called D-glucose or dextrose (dexter=Latin for right). It is possible to make L-glucose (its mirror-image) by chemical synthesis.
The human body cannot use L-glucose. It tasts just as sweet, but no calories can be gained from it. It will be secreted by the kidneys, where it may do some damage in the long term, or part of it may cause fermentation in the intestines (flatulence).A more notorious example is thalidomide (aka softenon and other names), prescribed in the 50s and early 60s for pregnant women to deal with morning sickness (a.o.). The laboratory stuff that was tested contained only one stereo-isomer, but the industrial product also contained the other one, which led to great deformities in newborn babies.
-
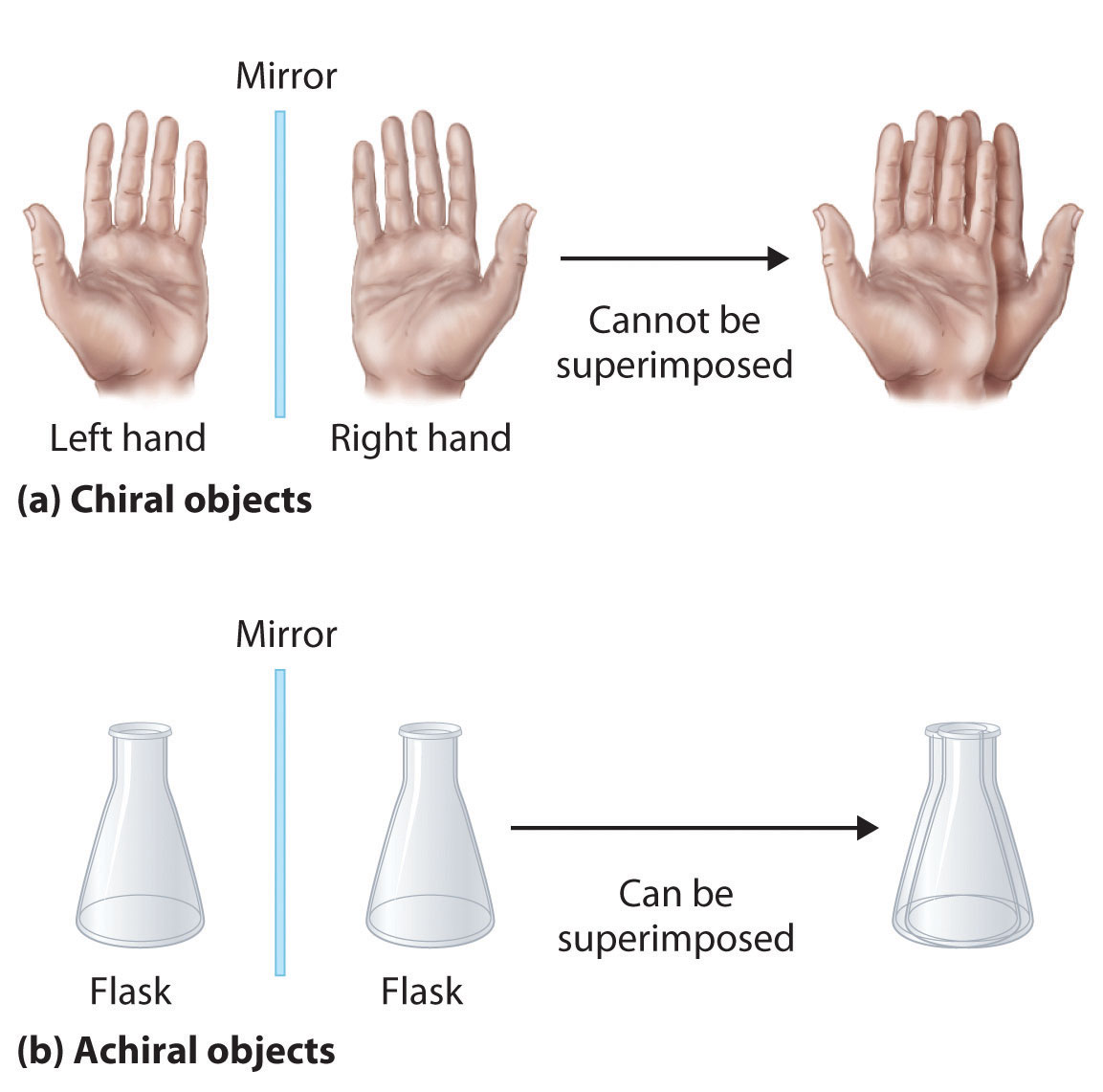
A molecule is considered chiral if there exists another molecule that is of identical composition but which is arranged in a non-superposable mirror image. Also the presence of an asymmetric carbon atom is often the feature that causes chirality in molecules.
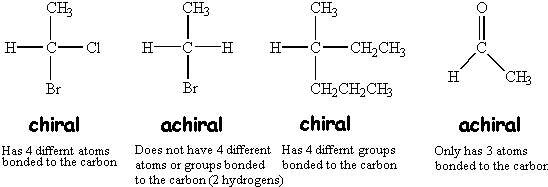
Two mirror images of a chiral molecule are called enantiomers or optical isomers. Pairs of enantiomers are often designated as "right-" and "left-handed".

Molecular chirality is of interest because of its application to stereochemistry in inorganic chemistry, organic chemistry, physical chemistry, biochemistry, and supramolecular chemistry.
-
Look for
#sp^3# carbons; those tend to be the most prevalent indications of chirality or achirality. You can ignore many#sp^2# alkene carbons.Note that only after examining a potential chiral carbon can you determine any achirality it actually has.
After you determine what atoms are stereocenters, if there is an even number of stereocenters, check whether or not the molecule has a symmetry axis dividing down the middle of the stereocenters.
If so, you might have a meso isomer, in which case the molecule as a whole is NOT chiral.
Here is an example of identifying stereocenters in detail:
http://socratic.org/questions/can-someone-please-explain-this-to-me-how-to-identify-the-stereo-centers-as-well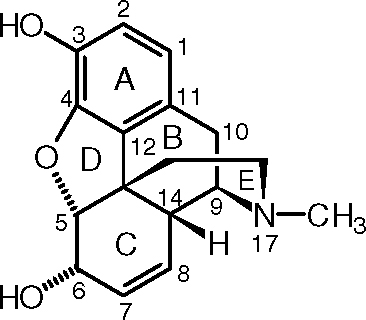
Can you see that carbons 5, 6, 9, 13, and 14 are stereocenters? Can you identify which configuration they are?
When you figure it out, check out the link above and see what they actually are.