Octet Rule
Key Questions
-
But of course...
and we see that in many molecules, like the trigonal bipyramidal
#"PCl"_5# , the octahedral#"SF"_6# , and the square pyramidal#"ClF"_5# . There are many other examples among transition metal complexes that I won't mention.Here,
#10# valence electrons surround phosphorus.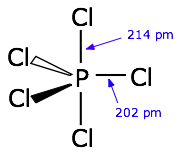
Here,
#12# valence electrons surround sulfur.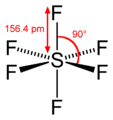
Here,
#12# valence electrons surround chlorine.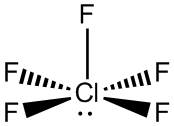
This is possible because all three central atoms have access to orbitals on the third quantum level,
#n = 3# . In all of these cases, the#3d# orbitals can be used, allowing extra space to store more than#8# total valence electrons.